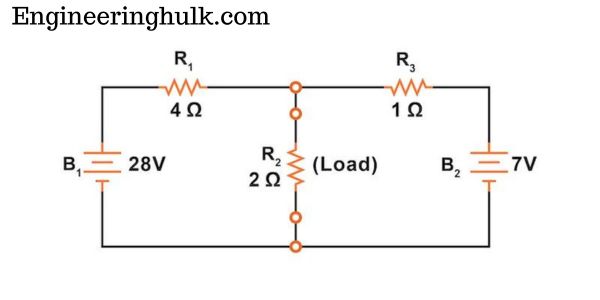
Table of Contents
Introduction
In the vast realm of science, understanding the fundamental nature of matter is a pursuit that has captivated scientists for centuries. One of the most crucial concepts in the realm of chemistry is Avogadro’s number.
What is Avogadro’s Number?
Avogadro’s number, denoted as “Nₐ,” is a fundamental constant in chemistry and physics that represents the number of atoms or molecules in one mole of a substance. It is named after the Italian scientist Amedeo Avogadro, who proposed this concept in the early 19th century. Avogadro’s number has been determined experimentally and has a value of approximately 6.022 x 10²³ particles per mole.
Understanding the Mole
Before delving deeper into Avogadro’s number, it is essential to grasp the concept of the mole. A mole is a unit of measurement used in chemistry to quantify the amount of a substance. One mole of any substance contains Avogadro’s number of particles, whether they are atoms, molecules, ions, or any other fundamental particles.
The mole allows scientists to work with macroscopic quantities of substances while dealing with the atomic and molecular scale.
Avogadro’s Number and the Atomic World
The significance of Avogadro’s number lies in its ability to bridge the gap between the macroscopic world we observe and the microscopic world of atoms and molecules. It provides a way to relate measurable quantities to the atomic and molecular scale.
For instance, consider the element carbon. Its atomic mass is approximately 12 atomic mass units (u). Avogadro’s number tells us that in 12 grams of carbon, there are precisely 6.022 x 10²³ carbon atoms. This remarkable relationship allows scientists to calculate the number of atoms or molecules in a given mass of any substance accurately.
Applications of Avogadro’s Number
Avogadro’s number finds widespread applications in various scientific and technological fields. Let’s explore a few key areas where Avogadro’s number plays a crucial role:
1. Stoichiometry:
In chemical reactions, stoichiometry determines the quantitative relationship between reactants and products. Avogadro’s number allows scientists to establish the ratio of atoms or molecules involved in a reaction, facilitating precise calculations of reactants and products.
2. Molar Mass:
The molar mass of a substance is the mass of one mole of that substance. By using Avogadro’s number, scientists can determine the molar mass of elements and compounds, aiding in the understanding of their chemical properties and reactions.
3. Gas Laws:
Avogadro’s number is an essential component of gas laws, such as Avogadro’s law, which states that equal volumes of gases, at the same temperature and pressure, contain an equal number of particles. This concept provides a foundation for understanding the behavior of gases and helps in various applications, including gas storage and compression.
4. Nanotechnology:
In the emerging field of nanotechnology, where materials and devices are manipulated at the nanoscale, Avogadro’s number is crucial. It helps scientists understand and work with extremely small quantities of atoms and molecules, enabling advancements in nanomaterials, nanoelectronics, and nanomedicine.
Determining Avogadro’s Number: Experimental Methods:
Various experimental techniques have been developed to determine Avogadro’s number. Here are some notable methods:
a. Measurement of Electrolysis:
In the late 19th century, the electrolysis of silver and copper salts was used to determine the amount of substance in terms of electrons transferred. By accurately measuring the charge passed during electrolysis, Avogadro’s number could be calculated.
b. X-Ray Crystallography:
This method involves analyzing the atomic arrangements within crystals. By measuring the volume occupied by each atom in the crystal lattice, scientists can indirectly determine Avogadro’s number.
c. Sedimentation and Diffusion:
French physicist Jean Perrin conducted groundbreaking experiments in the early 20th century using the sedimentation and diffusion of small particles in a liquid. These techniques allowed for the determination of Avogadro’s number based on statistical analysis and particle movement.
d. Silicon-28 Approach:
In recent years, a highly accurate determination of Avogadro’s number was achieved using a single crystal of highly enriched silicon-28 atoms. By counting the number of atoms in a precisely measured silicon sphere, scientists obtained a highly precise value for Avogadro’s number.
The Role of Technology and Innovation:
Advancements in technology have significantly contributed to the accuracy of Avogadro’s number determination. Modern techniques, such as laser interferometry and ultra-precise measurement devices, have enhanced the precision and reliability of experimental methods.
Also, read Newtons second law





























Comment on “Avogadro’s Number: Unlocking the Secrets of the Atomic World”
Comments are closed.