In the past, a great deal of heat treatment, mainly of tools, was done in a blacksmith’s fire, and even today this method is still used in some places. At best, however, this heating method is unreliable, as for all but small tools the heating is not uniform and, more importantly, estimating the correct temperature depends on the skill and experience of the user blacksmith.
If the steel is too hot, it burns, and if the critical range is not reached, the changes sought in treatment do not occur. Ovens are made in many shapes, sizes and varieties. Heating methods are generally by coal, oil, gas or electricity, and ovens can be obtained in capacities ranging from small chambers measuring about 150 mm wide X 100 mm high X 200 mm deep, suitable for small tools, to huge structures many meters long for the heat treatment of large bars and forgings. Salt bath ovens.
For some purposes, mainly for the treatment of tools and special steels, ovens are used that have a molten salt bath as a heating method. For example, sodium cyanide melts and melts at about 600°C, and in its molten state, it can be heated to about 900°C. If therefore, we wish to heat certain articles to temperatures between these limits, an excellent method of doing so is to dip them in a bath of molten cyanide until they reach their temperature.
As long as they are immersed in the liquid salt, they are protected from the air and therefore do not oxidize and encrust and, moreover, are heated evenly on all sides. Ovens of this type are called salt bath ovens.
The use of these ovens involves taking certain precautions against the fumes emitted, and care must be taken when tempering items that may have a coating of molten salt because of the spitting that may occur. To protect against this second risk, operators often wear protective gloves and eyewear.
A corner of a heat treatment shop is shown in Figure, the furnaces being (a) and (b) gas (or muffle) furnaces, (c) small tempering furnace with a separate metal vessel, and (d) furnace heated to gas salt bath oven (shown with doors closed).

Furnace temperatures:
An important aid to a furnace is some method of measuring its temperature because successful heat treatment of steel depends on close adherence to the correct temperature. There are many methods used for this, being a simple one to put in the oven some substance that melts at the temperature you want to check.
The substances used for this are moulded in the form of cones of mixtures of kaolin, lime, feldspar, magnesia, quartz and boric acid, with their melting temperatures arranged in stages from 600 °C to 2000 °C.
When a furnace temperature is required, several such cones, covering a range of melting temperatures with which the furnace temperature is considered, are placed and observed. The temperature is then judged from which cones collapse and which remain unaffected by the heat of the oven.
For example, to verify an assumed temperature of 810°-820°C, cones with melting points of 790, 815 and 835°C can be placed, the temperature is estimated from their condition after sufficient time has elapsed for them to be affected. These cones are called Seger Cones or Sentinels.
Pyrometers
For modern heat-treating furnaces, the above method of temperature measurement is not very convenient because it is time-consuming in operation and does not provide a continuous temperature reading as is often required. A more scientific and reliable method of measuring oven temperatures is with an instrument called a pyrometer. There are several forms of pyrometers and two types in common use are:
(1) The Thermo-Electric Pyrometer
(2) The Optical Pyrometer.
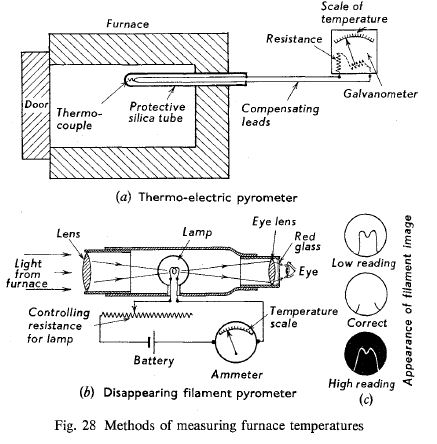
The first type uses the principle that when two different wires are joined to form a complete electrical circuit, and the two junctions are kept at different temperatures, an electric current flows in the circuit, the magnitude of the current depending on the metals used, and the difference in junction temperature. The hot junction, which is placed in the oven, is usually made up of platinum wires and a soldered platinum-rhodium alloy and is called a thermocouple.
The conductors of these wires are brought to a sensitive galvanometer which usually constitutes the cold junction, and which indicates the current flowing in the circuit due to the temperature difference between the two junctions. The galvanometer is so calibrated that instead of indicating electrical units, it reads in degrees of temperature. A diagram of this pyrometer is shown in Fig. 28(a).
The optical pyrometer compares the intensity of the light emitted by the oven with that of some standard source. In the fading filament pyrometer, the brightness of a standard filament bulb is varied until it matches the oven light and disappears when viewed through the telescope. The instrument, which is schematically shown in Fig. 28(b), is placed in front of the oven and the light from the oven is seen through the eyepiece.
The current through the lamp is varied by resistance, and when a colour match is obtained the lamp filament disappears from view, the lamp current being necessary for this to be indicated on an ammeter, this instrument can be calibrated so that it reads in degrees of temperature, rather than in units of electric current.